
The UF/IFAS Indian River Research and Education Center, with Rising Tide Conservation, is pleased to announce the successful aquaculture of the copperband butterflyfish, Chelmon rostratus!
This success was achieved by the hard work of Dr. Cortney Ohs, Katie McCord (RTC funded MS student), Morgan Bronson (MS student), Audrey Beany (Biologist), Peter Woodward (Live Feeds Technician), and John Marcellus (Aquaculture Systems Maintenance), with research funding from Rising Tide Conservation.

Family: Chaetodontidae (Butterflyfish)
Genus: Chelmon
Common names: Copperband Butterflyfish, Copperbanded Butterflyfish, Beaked Butterflyfish, Long-nosed Butterflyfish, Beaked Coralfish, Orange-Stripe Butterflyfish
Copperband butterflyfish, Chelmon rostratus, can be found in the tropical Indo-Pacific, extending from Japan’s Ryuku Islands, the Andaman Sea across to Papua New Guinea, and along much of Australia’s northeastern/eastern coast.
Copperbands can reach lengths of around 20 cm (8 in) and have dramatic, distinct coloring with coppery, orange/yellow vertical bars set against a pearlescent white body. They have a black false eyespot outlined in blue, and a black band at the base of the caudal fin. Their prominent snout allows them to reach into small reef crevices for their prey, which includes anemones (Aiptasia sp.), polychaete worms, small crustaceans, and occasionally coral polyps.
They inhabit rocky and coral reefs down to depths of 25 m (~82 ft), though they commonly occur at shallower depths. Copperbands are territorial, and adults are found singly or in monogamous breeding pairs. There is some sexual dimorphism, males’ snout has “a small hump…and higher slope than female(s).”¹
Aquaculture History
In 2016, Ruensirikul, Jirayuth, et al., published their work successfully pairing and spawning captive copperband butterflyfish, and larval rearing to 10 days post hatch (DPH). No other reports were found regarding captive spawning or larval rearing of copperbands.

Aquaculture Process
Broodstock Conditioning
Copperband butterflyfish broodstock pairing attempts started in 2017 with one successful pair, then additional potential broodfish were obtained in 2018. The pairing process began after the quarantine period. The fish did not show sexual dimorphism at the time they were acquired, so pairs, trios, and groups of four were stocked into shallow blue poly tanks. These tanks initially had clear dividers so the fish could see each other but were not in direct contact, and their behavior was observed closely over a few months. From these observations potential pairs were combined together. Bonded pairs were determined by additional behavior observations, with successful pairs following each other, swimming side by side frequently, and not displaying aggression. Individuals that did not form a bonded pair were separated and then attempted to be paired to other fish. Broodstock development took about two years once paired. Pairing occured when the fish were 5 – 6.5 cm (~2 – 2.5 in) in length, and spawning occured when the pair reached about 10 – 13 cm (~ 4-5 in) in length.
Currently, there are four bonded pairs of copperband butterflyfish at IRREC. One pair (henceforth referred to as “Pair A”), consisting of the largest-sized individuals, is spawning consistently. Another pair has spawned a few infertile eggs but is not consistently spawning yet. Two other pairs may not be sexually mature yet.
Spawning Broodstock “Pair A” Profile
Time Paired at First Spawn: 2 years
Size at First Spawn: 10 – 13 cm (~ 4-5 inches) long
Housing: 1600L tank system, with cone top cover and a heat pump which maintains temperature at 25.5℃ +/- 1℃ (77.9℉).
Light cycle: 14 hours of direct light with 3 hours of sunrise and sunset by ambient building lights.
Diet: Frozen squid, frozen krill, frozen PE mysis shrimp from Piscine Energetics, and LRS Fertility Frenzy
Conditions at First Spawn:
Water Temp: 25.5℃ (77.9℉)
Salinity: 34 g/L
Light cycle: 14 hours light / 10 hours dark

Spawning and Egg Collection
Broodstock Pair A has been spawning two days in a row every two or three weeks for over a year. Initially, eggs were observed to be fertilized but the embryo did not develop properly. The improper development looks like a flower petal in the egg, so division is happening but development was ceasing at about the 32 to 64 cell stages for an unknown reason. This improper development has also been observed in different species of butterflyfish being studied at the lab. Collected eggs were held in a bucket with clean water and aerated and no further development occurred. Some eggs floated while others sank.
After multiple, consistent spawns containing eggs with improper embryo development, a full water change on the tank was timed to be a day or two before the next expected spawn. A 95% water change was done without removing the fish from the tanks, they were allowed to swim in the remaining water. The water was replaced with fresh, sterile Atlantic Ocean water (Salinity: 34 g/L and Temp: 25℃/77℉). The first spawn after the water change contained the first fertilized eggs with proper embryo development. The only tested water quality parameter that was out of preferred range was nitrate. The nitrate was >160 mg/L before the water change, and 10 mg/L after. It is unclear if it was the high nitrate or another ion was high or possibly depleted which influenced the improper embryo development, but a full water change allowed for proper embryo development.
Eggs were collected in upwelling egg collectors and were always removed in the morning before feeding, then were held in 1 L containers with aeration. Quantity of fertilized and unfertilized eggs were estimated by counting triplicate volumetric samples under a dissecting microscope. It was observed that after holding the fertilized eggs, some would continue to develop into a proper embryo by late in the afternoon. This indicated that either the embryos develop slower than other species of fish (18-24 hours) or they spawned near dawn instead of sunset like most of the other species of fishes at the lab. We now believe the copperband broodstock Pair A spawns at pre-dawn or first light, similar to milletseed butterflyfish, Chaetodon miliaris, and the copperband eggs have a standard 18-24 hour embryo development.
Between August 2020 to April 2021, spawning by broodstock Pair A was recorded 25 times (180-55,000 eggs/spawn). Of these 19 spawns were only unfertilized eggs, while six had partially fertilized eggs (0.12-85.2% fertilization).
Larval Development
The weekend of February 27, 2021, two spawns from Pair A were recorded, consisting of 55,186 eggs (3.98% fertilization) and 22,880 eggs (32.25% fertilization) with a hatch rate estimated around 10%. Newly hatched larvae are described as strong fast swimmers but small, a similar size to Pacific blue tang, Paracanthurus hepatus, larvae.
Copperband larvae are housed in 29L fiberglass tanks, which have a white bottom and black walls. All tanks are part of the larger recirculating aquaculture system serving all larval tanks. Temperature is maintained at 25-26℃ (77-79℉) by room temperature control.
Larvae At Hatch:
Size: 2 mm
Yolk Sac Present: Yes
Mouth Present: No
Eyes: Present but not pigmented, maybe not functional
3 DPH (Days Post Hatch): Size: 2.4 mm. Less than half of the larvae had mouth parts. First feeding.
8 DPH: Swim bladder inflation.
Approx. 8 DPH- 30 DPH: Tholichthys stage. Larvae have notably short but deep bodies as compared to many other species of marine fish larvae. They have helmet-like head plates with 2 peaks, long operculum spikes, a small mouth, round caudal fin, and the dorsal fin is short in front and taller further towards the tail.
Approx. 20 DPH: Flexion occured.
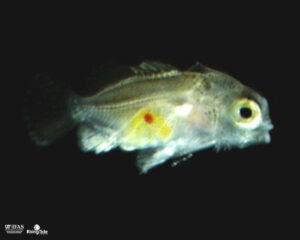
51 DPH: Coloration changes noticeable, dark (black) diffuse vertical bands and black eyespot present on some.
53 DPH: The eyespot has a partial copper ring forming and black bands are highly visible.
57 DPH: Copper bands visible within the black bands on the anterior half of the body and eyespot is encircled in a copper ring.
Approx. 60 DPH: Settlement occurred. Larvae are housed in 29L tanks which makes it difficult to determine exactly when settlement occurred. Settlement was determined by behavior, at 60 DPH larvae were observed to be benthically oriented, only coming to the surface to feed on adult copepods.











Larval Feeding/ Daily Feeding Protocol
Live Tisoschrysis lutea algae added daily before lights turned on at 200-300,000 cells/mL.
Aeration was moderate with a small airstone along the wall of the tank.
Lighting was fluorescent lights providing 750 lux of lighting at the water surface.
Flow thru water was turned off and tanks were left stagnant with only aeration from lights on to lights off. Then water was flushed throughout the entire night to attempt to remove uneaten copepods.
Screen mesh size on the drains increased as the larvae grew; they were 150 micron for the first 7 DPH, then 300 micron until 30 DPH, and finally were increased to 500 micron.
3 DPH: First day of feeding, 5/mL of parvo and rotifers once daily.
4 DPH: Fed the same, once daily.
5 DPH: Fed the same, twice daily.
6-8 DPH: Fed 10/ml twice daily for both parvo and rotifers.
9-16 DPH: Continued with same feeding rates.
17-26 DPH: Fed 10/mL in morning and 10-17/mL in afternoon.
27-36 DPH: Fed 15/mL twice daily.
37 DPH: Fed 15/mL in morning and adult copepods.
38-41 DPH: Fed 15/mL twice daily and adult copepods.
42-50 DPH: Fed 15/mL in morning 10/mL in afternoon and adult copepods maintained.
Adult copepods were added ad libitum and maintained at a visible presence density.

Sources
- Chelmon rostratus, Copperband butterflyfish via FishBase
- Myers, R. & Pratchett, M. 2010. Chelmon rostratus. The IUCN Red List of Threatened Species 2010: e.T165659A6083324. https://dx.doi.org/10.2305/IUCN.UK.2010-4.RLTS.T165659A6083324.en.
- Revisited: Copperband Butterflyfish via Quality Marine
- Ruensirikul, Jirayuth, et al. “Sexual dimorphism identification, response to pairing manipulation and spawning in captivity of copperband butterflyfish, Chelmon rostratus (Linnaeus, 1758).” Thai Fisheries Gazette (Thailand) (2016).
- The long-nosed butterflies part 1: Chelmon and Chelmonops via Reefs.com



